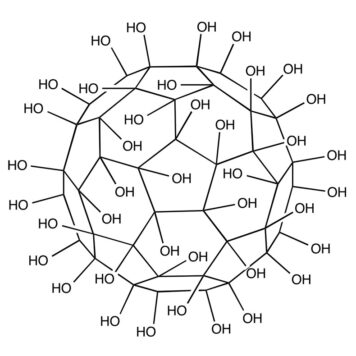
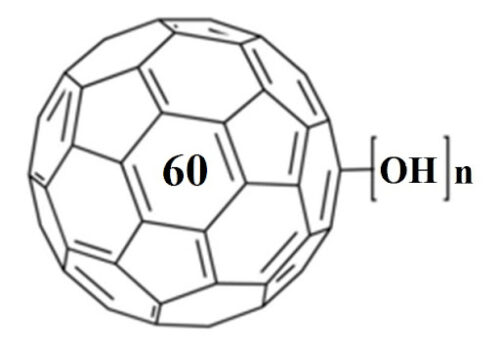
The surface modification of commercial TiO2#nbsp;Hombikat (TiO2) using nanoparticles of fullerene C60#nbsp;with tetrahydrofuran (THF-nC60), as well as fullerenol C60(OH)24#nbsp;nanoparticles (FNP) was investigated in this study. Characterization of THF-nC60, FNP, TiO2, TiO2/THF-nC60, and TiO2/FNP was studied by using DES, ELS, TEM, SEM, DRS and BET measurements and their photoactivity has been examined on the mesotrione degradation under simulated sunlight. It was found that FNP in self-assembled nanocomposite TiO2/FNP increased negatively charge, as well as catalytic surface of TiO2. In addition, TiO2/FNP exhibits a shift of band gap energy to lower values compared to TiO2#nbsp;and TiO2/THF-nC60. BET surface area has not showed significant differences among catalysts. Furthermore, it was found that the highest photoactivity was obtained for TiO2/FNP system. Besides, influence of different concentrations of electron acceptors (H2O2#nbsp;and KBrO3), as well as scavengers on the kinetics of mesotrione removal in aqueous solution with/without TiO2#nbsp;and FNP under simulated sunlight was investigated. Namely, addition of mentioned electron acceptors has resulted in higher mesotrione degradation efficiency compared to O2#nbsp;alone. Besides, in the first period substrate degradation probably takes place via hydroxyl radicals and after 60 min of irradiation the reaction mechanism proceeds mainly via holes. The most efficient system for mesotrione degradation and mineralization were TiO2/7 mM KBrO3#nbsp;and TiO2/7 mM KBrO3/40 μl FNP, respectively.
Related researches 71 articles














![Inhalable gadofullerenol/[70] fullerenol as high-efficiency ROS scavengers for pulmonary fibrosis therapy](https://biofullerene.com/wp-content/uploads/2022/12/istockphoto-12925559-440x356.jpg)

















![Palladium-Catalyzed Reaction of [60]Fullerene with Aroyl Compounds via Enolate-Mediated sp 2 C-H Bond Activation and Hydroxylation](https://biofullerene.com/wp-content/uploads/2022/12/2978543-356x356.png)