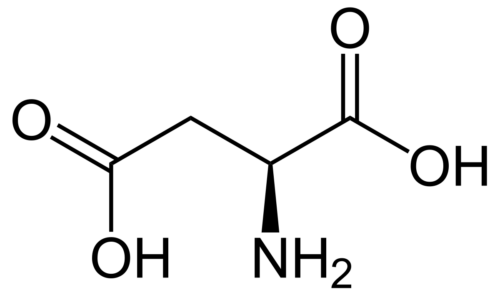
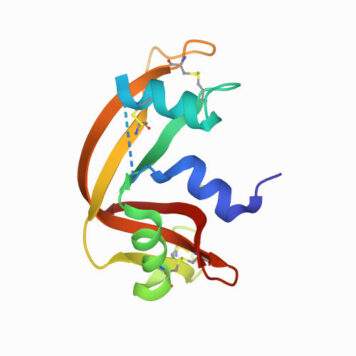
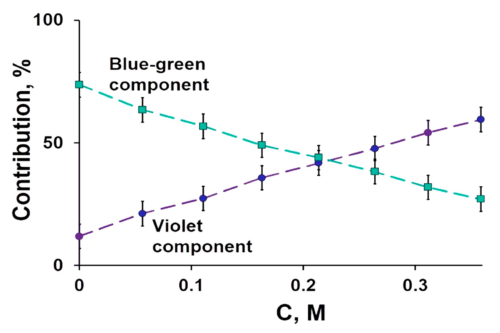
Protein ubiquitination plays a role in essentially every process in eukaryotic cells. The attachment of ubiquitin (Ub) or Ub-like (UBL) proteins to target proteins is achieved by parallel but distinct cascades of enzymatic reactions involving three enzymes: E1, E2, and E3. The E1 enzyme functions at the apex of this pathway and plays a critical role in activating the C-terminus of ubiquitin or UBL, which is an essential step that triggers subsequent downstream transfer to their cognate E2s resulting in the fidelity of the Ub/UBL conjugation machinery. Despite the central role of the E1 enzyme in protein modification, a quantitative method to measure Ub/UBL activation by E1 is lacking. Here, we present a mass spectrometry-based assay to accurately measure the activation of Ub/UBL by E1 independent of the E2/E3 enzymes. Our method does not require radiolabeling of any components and therefore can be used in any biochemical laboratory having access to a mass spectrometer. This method allowed us to dissect the concerted process of E1-E2-catalyzed Ub conjugation in order to separately characterize the process of Ub activation and how it is affected by select mutations and other factors. We found that the hydrophobic patch of Ub is important for the optimal activation of Ub by E1. We further show that the blockers of the Ub-proteasome system such as ubistatin and fullerenol inhibit Ub activation by E1. Interestingly, our data indicate that the phosphorylation of Ub at the S65 position augments its activation by the E1 enzyme.
Related researches 41 articles







![Biocompatible [60]/[70] Fullerenols: Potent Defense against Oxidative Injury Induced by Reduplicative Chemotherapy](https://biofullerene.com/wp-content/uploads/2022/11/istockphoto-65584859-356x356.jpg)