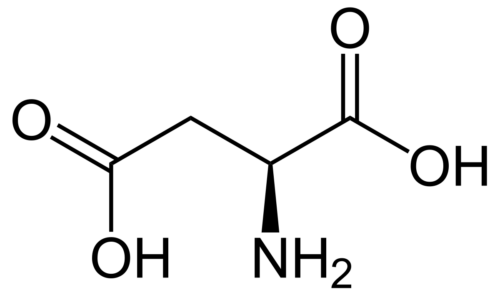
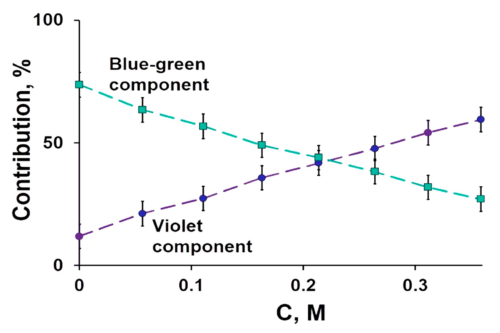
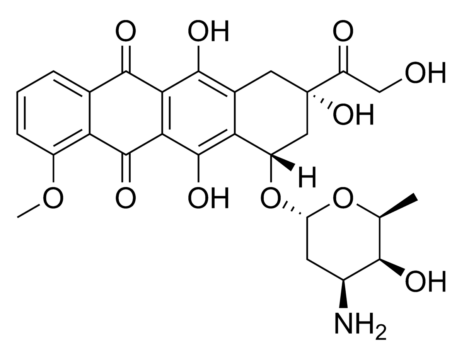
Derivatization of fullerenes to polyhydroxylated fullerenes, i.e., fullerenols (FLU), dramatically decreases their toxicity and has been reported to enhance the solubility as well as cellular permeability. In this paper, we report synthesis of FLU as nanocarrier and subsequent chemical conjugation of Methotrexate (MTX) to FLU with a serum-stable and intracellularly hydrolysable ester bond between FLU and MTX. The conjugate was characterized for physiochemical attributes, micromeritics, drug-loading, and drug-release and evaluated for cancer cell-toxicity, cellular-uptake, hemocompatibility, protein binding, and pharmacokinetics. The developed hemocompatible FL-MTX offered lower protein binding vis-à-vis naïve drug and substantially higher drug loading. The conjugate offered pH-dependent release of 38.20 ± 1.19% at systemic pH and 85.67 ± 3.39% at the cancer cell pH. FLU-MTX-treated cells showed significant reduction in IC50#nbsp;value vis-à-vis the cells treated with pure MTX. Analogously, the results from confocal scanning laser microscopy also confirmed the easy access of the dye-tagged FLU-MTX conjugate to the cell interiors. In pharmacokinetics, the AUC of MTX was enhanced by approx. 6.15 times and plasma half-life was enhanced by 2.45 times, after parenteral administration of single equivalent dose in rodents. FLU-MTX offered enhanced availability of drug to the biological system, meanwhile improved the cancer-cell cytotoxicity, sustained the effective plasma drug concentrations, and offered substantial compatibility to erythrocytes.
Related researches 41 articles








![Biocompatible [60]/[70] Fullerenols: Potent Defense against Oxidative Injury Induced by Reduplicative Chemotherapy](https://biofullerene.com/wp-content/uploads/2022/11/istockphoto-65584859-356x356.jpg)