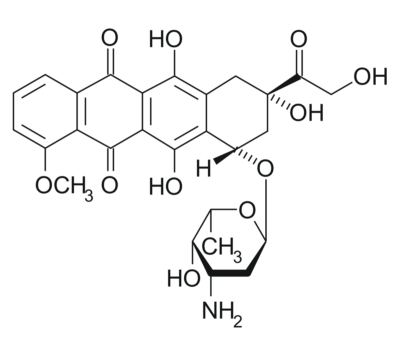
Polyhydroxylated fullerenes (fullerenols) are excellent free radical scavengers. Despite the large number of reports on their reactions with reactive oxygen species, there is no report on their ability to trap lipid peroxyl radicals and act as chain-breaking antioxidants. In this work we studied the effect of fullerenol C60(OH)36 on the kinetics of peroxidation of polyunsaturated fatty acid ester (methyl linoleate) dispersed in two model systems that mimic biological systems: Triton X-100 micelles and Large Unilamellar Vesicles, at pH 4, 7 and 10. As a control antioxidant 2,2,5,7,8-pentamethyl-6-hydroxychroman (PMHC, an analog of α-tocopherol) was used. In micellar systems at pH 4.0, C60(OH)36 reacts with peroxyl radicals with kinh= (5.8 ± 0.3) × 103 M−1s −1 (for PMHC kinh = 22 × 103 M−1s −1). Surprisingly, at pH 7 a retardation instead of inhibition was recorded, and at pH 10 no effect on the kinetics of the process was observed. In liposomal systems fullerenol was not active at pH 4.0 but at pH 7.0 kinh = (8.8 ± 2.6) × 103 M−1s −1 for fullerenol was 30% lower than kinh for PMHC. Using two fluorescent probes we confirmed that at pH 7.4 fullerenol/fullerenol anions are incorporated into the phospholipid heads of the bilayer. We also studied the cooperation of C60(OH)36 with PMHC: both compounds seem to contribute their peroxyl radical trapping abilities independently at pH 4 whereas at pH 7 and 10 a hypersynergy was observed. The antioxidant action of C60(OH)36 and its synergy with PMHC was also confirmed for peroxidation of human erythrocytes at pH 7.4. Assuming the simplified structural model of fullerenol limited to 36 hydroxyls as the only functional groups attached to C60 core we found by density-functional theory a low energy structure with OH groups distributed in the form of two polyhydroxyl regions separating two unsubstituted carbon regions with biphenyl-like structure. Our calculations indicate that abstraction of hydrogen atom from fullerenol by peroxyl or tocopheroxyl radical is endoergic. As the electron transfer from fullerenol polyanion to the radicals is also energetically disfavoured, the most probable mechanism of reaction with radicals is subsequent addition of peroxyl/tocopheroxyl radicals to biphenyl moieties surrounded by OH groups.
Related researches 23 articles