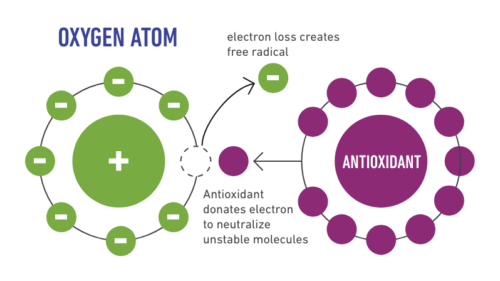
Antioxidant ability of the water-soluble derivative of fullerene (C60), prepared by high-degree hydroxylation [C60-(OH)(32) x 8H(2)O] or C60/gamma-cyclodextrin (1:2 mol/mol) clathrate formation [C60/(gamma-CD)(2)], was assessed by electron spin resonance method and beta-carotene bleaching assay. These C60 derivatives have an ability to diminish a 1:2:2:1 quartet ESR spectrum attributed to hydroxyl radicals ((.)OH) as shown by DMPO-spin trap/ESR method. Meanwhile, a singlet radical-signal different from ()OH-attributed signals increased in a manner dependent on concentrations of C60-(OH)(32) x 8H(2)O. This might suggest that C60-(OH)(32) x 8H(2)O scavenges (.)OH owing to dehydrogenation of C60-(OH)(32) x 8H(2)O, and is simultaneously oxidized to a stable radical species, which may be a dehydrogenated fullerenol radical (C60-O(.)). Furthermore, these water-soluble derivatives of C60 suppressed fading of yellowish color characteristic of intact beta-carotene in beta-carotene bleaching assay. Antioxidant abilities of these derivatives were assessed as retention of yellowish color (viz absorbance at 470 nm) for 180 min. Namely, beta-carotene-attributed chromaticity (% relative absorbance at 470 nm compared with the control) after 180 min was 69% for C60-(OH)(32) x 8H(2)O (400 microM: C60-eq.), and 32% for C60/(gamma-CD)(2) (400 microM: C60-eq.), whereas it was 6% for l(+)-ascorbic acid (400 microM) which is hydrophilic, and 85% for (+/-)-alpha-tocopherol (400 microM) which is lipophilic, respectively. Thus C60-(OH)(32) x 8H(2)O and C60/(gamma-CD)(2) can scavenge (.)OH, and have a distinct antioxidative activity in the aqueous system containing linoleic acid which is abundantly contained in the cell membrane together with other unsaturated lipids. These C60 derivatives have a potential to protect the cell membrane from oxidative stress due to (.)OH.
Related researches 23 articles